Together Is Better: The Rumen Microbial Community as Biological Toolbox for Degradation of Synthetic Polyesters
- 1Department of Agrobiotechnology IFA-Tulln, Institute of Environmental Biotechnology, University of Natural Resources and Life Sciences, Vienna, Austria
- 2Division Polymers & Enzymes, Austrian Centre of Industrial Biotechnology, Graz, Austria
- 3Institute of Microbiology and Microbial Biotechnology, University of Natural Resources and Life Sciences, Vienna, Austria
- 4Department of Material Sciences and Process Engineering (MAP), Institute of Physics and Materials Science (IPM), Vienna, Austria
- 5Department of Microbiology, University of Innsbruck, Innsbruck, Austria
Microorganisms, like bacteria and fungi, are becoming an emerging resource for the development of eco-sustainable plastic degradation and recycling processes. In this study, the rumen content from cattle (Bos taurus) was investigated regarding synthetic polyester hydrolyzing enzymes based on the fact that the diet of ruminants may contain natural plant polyesters. A screening with model substrates demonstrated hydrolytic activities of rumen fluid on p-NP-esters with four to eight carbon atoms. Rumen fluid hydrolyzed synthetic aromatic polyesters with higher amounts of terephthalic acid released from poly(butylene adipate-co-terephthalate) (PBAT) (0.75 and 0.5 mM for polymer powder and film, respectively) and thus exceeded when compared to the hydrolysis of the second terephthalic acid-based polymer—poly(ethylene terephthalate) (PET) (0.6 and 0.15 mM, for powder and film, reciprocally). Additionally, rumen fluid hydrolyzed the bio-based polyester poly(ethylene furanoate) (PEF) according to HPLC and SEM analysis. Shotgun metagenome analysis of the rumen microbiome revealed the real proportion of all domains of life, showing the dominance of bacteria (98%), followed by Eukaryota (1%) and finally Archaea. Among the most abundant genera encountered in this study, polyester hydrolysis activity has already been proven (e.g., Pseudomonas).
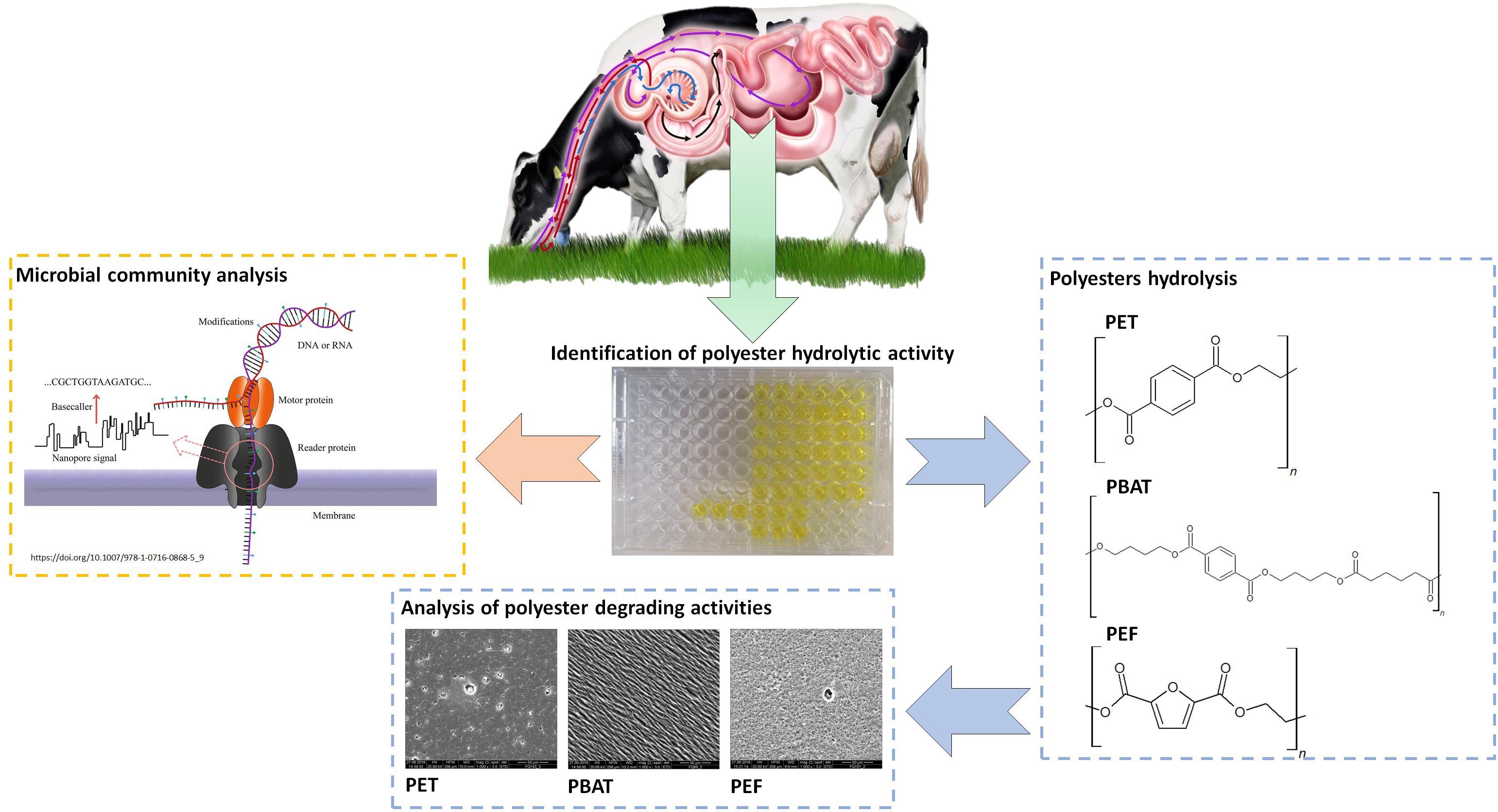
Graphical Abstract. Identfication of plastics degradation and microbial community analysis of Rumen.
Introduction
In Europe, the widespread consumption of plastic waste led to the accumulation of 25.8 million tons of waste in the terrestrial and marine environment, with polyesters accounting for approximately 15% thereof (Plastics Europe and EPRO, 2019). Recently, increasing public awareness and changing legislation have pushed the development of new strategies to reduce this huge environmental problem. Moreover, in the last two decades, the concept of bio-economy motivated the scientific community to focus on production of bio-based and/or biodegradable polyesters and/or technologies for microbial or enzymatic recycling (Pellis et al., 2016b). Thereby, the application of highly specific enzymes allows for a stepwise recovery of highly valuable building blocks from blended plastics or mixed waste streams. Previous studies from our group and others have demonstrated the potential of enzymes for the hydrolysis of poly(ethylene terephthalate) (PET), the most important synthetic polyester used in numerous applications including textiles and packaging (Herrero Acero et al., 2011; Pellis et al., 2016b; Quartinello et al., 2017). Apart from PET, two further polyesters recently gaining interest were assessed in this study, namely, the biodegradable poly(butylene adipate-co-terephthalate) (PBAT) and the bio-based poly(ethylene furanoate) (PEF). PBAT is a copolymer consisting of an aliphatic part (units of adipic acid and 1,4-butanediol) and the aromatic unit (terephthalic acid) (Biundo et al., 2016). Thanks to its properties, PBAT finds applications mostly as food packaging material and compostable plastic bags and, as a result of the presence of the aliphatic chains, renders the material compostable. On the other hand, PEF is based on 2,5-furandicarboxylic acid which is produced from renewable resources (Weinberger et al., 2017a). Several studies defined that mechanical properties (e.g., gas barrier of oxygen, carbon dioxide, and water vapor) of PEF are comparable to PET, while the bio-based origin can drastically reduce greenhouse emissions (Weinberger et al., 2017a). While enzymatic hydrolysis of these polyesters has been reported, there is still a strong demand for more efficient enzymes allowing industrial implementation. In addition to cultivation-based (Yoshida et al., 2016) or in silico-based screening of polyester-degrading microorganisms and their enzymes eventually followed by genetic engineering (Biundo et al., 2018), metagenomic approaches allow for the assessment of enzymes produced by microorganisms living almost in any niche in the biosphere with distinct roles in the ecosystem.
Using the above approach, microbial polyester hydrolyzing enzymes have been identified from different ecosystems including leaf-branch compost (Sulaiman et al., 2012), marine (Pinnell and Turner, 2019), and moss-associated environments (Müller et al., 2017). The identified enzymes are mainly microbial cutinases able to hydrolyze the natural plant polyester cutin, a major component of the plant cell wall.
Following from metagenomic screening, different hydrolases with polyester-degrading abilities have been isolated from Actinobacteria (e.g., Thermobifida spp.) and from fungi (Fusarium solani, Humicola insolens, Aspergillus spp., and Rhizopus spp.) (Ribitsch et al., 2013; Wei and Zimmermann, 2017). Subsequently, some of the novel hydrolases were engineered to further improve their activities on synthetic polyesters (Ribitsch et al., 2013). However, many ecosystems still remain to be exploited for polyester-hydrolyzing activities.
Ruminants are a well-studied group of herbivorous mammals. They have evolved a forestomach, the rumen (or more precisely the reticulorumen), which is a large chamber (50–100 L capacity in adult cattle) where the ingested feed is subjected to microbial degradation before the real animal digestion. The content in the rumen is quite heterogeneous, consisting of a mass of float (forage), sediment, liquids, and microbial cells (Gijzen et al., 1987). The rumen ecosystems, in this sense, can be defined as a “chemostat” where different biochemical reactions are occurring. Among these, enzymatic hydrolysis, e.g., of (hemi)cellulose and the responsible enzymes have been studied intensively, while other polymer-degrading enzymes potentially present in rumen remained under-explored. Yet the diet of ruminants may contain natural plant polyesters (cutin), and thus the enzymes with hydrolytic activities may be present in the rumen (Mao et al., 2013). As a consequence, the main objective of this study therefore was to explore the microbial community of the rumen from ruminant animals regarding polyester-degrading enzyme activities.
Materials and Methods
Chemicals and Substrates
Buffer components, bovine serum albumin (BSA), para-nitrophenyl-esters (p-NP-esters), terephthalic acid (Ta), and formic acid were purchased from Sigma-Aldrich (United States). Fresh rumen liquid was obtained from a slaughterhouse (Mayr, Natters, Austria), which originated from an alpine pasture-fed young ox. Within 2 h of sampling, the rumen was sieved to <2 mm and immediately stored at −20°C, till the start of the experiments.
Poly(ethylene terephthalate) (PET) powder and film were purchased from Goodfellow (United Kingdom). Polybutylene adipate-co-terephthalate (PBAT) powder and film were provided from BASF (Germany). Poly(ethylene furanoate) (PEF) powder and film were supplied by Corbion (Netherlands). Molecular weight (kDa) are listed in Supplementary Table 1. All other chemicals and reagents were purchased from Sigma-Aldrich and used at reagent grade, without further purification if not otherwise specified.
Esterase Activity Assay and Protein Concentration Assays
The pH in rumen ranges between 6 (in case of animal acidosis) and slightly higher than 7 under normal conditions (Mao et al., 2013). As the exposure to oxygen causes a fast rumen acidification, thawed rumen samples were diluted in 100 mM pH 7 potassium phosphate buffer with a ratio of 1:10 and stirred for 2 h at 150 rpm. Esterase activities were measured with different p-nitrophenyl (p-NP)-esters as substrates: p-NP-acetate (p-NPA, C2), p-NP-butyrate (p-NPB, C4), p-NP-hexanoate (p-NPH, C6), p-NP-octanoate (p-NPO, C8), p-NP-decanoate (p-NPD, C10), and p-NP-laureate (p-NPL, C12) as described by Biundo et al. (2016). Protein concentration was determined according to the Bio-Rad protein assay (Bio-Rad Laboratories GmbH, Vienna, Austria, catalyst no.: 500-0006, Austria) (Pellis et al., 2016a).
Enzymatic Hydrolysis of Polymer Powders and Films
Briefly, 5.0 mg of the polymer powders (PET, PBAT, and PEF) were incubated with 2.0 ml of rumen solution. Incubations were conducted for 24, 48, and 72 h in an orbital shaker set at 150 rpm and mimicking the rumen temperature (i.e., 40°C). Blank reactions were carried out in buffer. All reactions were performed in triplicates (Quartinello et al., 2017).
PET, PBAT, and PEF amorphous film samples of 0.5 × 1.0 cm were washed three times as previously described by Vecchiato et al. (2017). After washing, the films were incubated with 2 ml of sieved rumen samples for 24, 48, and 72 h. Incubations were performed with the same settings as the powder’s incubation. To test activity of various microorganisms on polyesters from Penicillium citrinum, Beauveria bassiana, and Pseudomonas pseudoalcaligenes were isolated and cultivated as previously described (Liebminger et al., 2007; Almansa et al., 2009). Following incubation, the culture supernatants were collected and centrifuged at 4°C and 3,700 rpm for 25 min (Eppendorf Centrifuge 5920 R) and thereafter incubated at the same conditions performed as earlier described for rumen solution.
High-Performance Liquid Chromatography (HPLC-DAD)
After enzymatic treatment of polyester powders/films, proteins were precipitated with ice-cold MeOH. Samples were centrifuged at 12,700 rpm at 0°C for 15 min. The resulting supernatant was filtered (0.45 μm PA filters) and filled in an HPLC vial (Quartinello et al., 2017).
HPLC coupled with reversed-phase column C18 was used for analysis of hydrolysis products using a H2O/MeOH/HCOOH gradient (Supplementary Table 2). The flow rate was set to 0.85 mL min–1, and the column was maintained at a temperature of 40°C. The injection volume was 10 μl. Detection of the analytes was performed with a photodiode array detector at a wavelength of 245 nm (for PET and PBAT) and 260 nm (for PEF) (Weinberger et al., 2017b).
For analysis of PET hydrolysis products, Ta, mono-2-hydroxyethyl terephthalate (MHET), and bis(2-hydroxyethyl)terephthalate (BHET) calibration curves were prepared, while Ta, mono(hydroxybutyl)terephthalate (BTa), and bis(4-hydroxybutyl) terephthalate (BTaB) calibrations were prepared for PBAT released product detection (Biundo et al., 2016). A standard 2.5-furandicarboxylic acid (FDCA) calibration curve was used for quantification of PEF hydrolysis products (Perz et al., 2016).
Scanning Electron Microscopy (SEM)
For the SEM investigations, a FEI Quanta 250 FEG (Thermo Fisher Scientific, Hillsboro, OR, United States) was used under high-vacuum conditions and a variable high tension from 5 to 10 kV. The micrographs were recorded with the Everhart-Thornley detector in secondary electron (SE) mode. The fracture surface was sputter-coated with a 10-nm-thin layer of gold in order to provide sufficient electrical conductivity (Quartinello et al., 2019).
Microbial Community Analysis
DNA Extraction
Total DNA was isolated from a 1 ml rumen sample. Debris and liquid fraction were briefly vortexed for sample homogenization, split in two aliquots, and thereafter centrifuged. The pellets were then resuspended in 100 μl 0.01 M phosphate buffer. The DNA extraction was performed using the FastDNA SPIN kit for soil (MP Biomedicals, Illkirch, France) according to the manufacturer’s recommendations. In order to determine DNA purity and concentration to match the Nanopore workflow’s requirement, the extracts were pooled and purified using the QIAamp Viral RNA Mini Kit (Qiagen). The DNA concentration was assessed by using the Qubit 2.0 fluorometer (Invitrogen Corporation), with the Qubit dsDNA BR Assay Kit.
Library Construction, Template Preparation, and Sequencing
A shotgun metagenome analysis was performed with the extracted DNA without any step of amplification of target sequences. To this end, DNA libraries were constructed following the “1D Genomic DNA by Ligation” guideline available in the Oxford Nanopore community using the Ligation Sequencing kit SQK-LSK109 and the Flow Cell Priming Kit EXP-FLP001 (Oxford Nanopore, United Kingdom). All steps for library construction were performed following the specifications of the protocol and using 1 μg input of extracted DNA. Following the preparation of the DNA ends for adapter attachment using the NEBNext FFPE DNA repair mix (M6630) and the NEBNext End Repair/dA-Tailing Module (E7546) (New England Biolabs), the attachment of sequencing adapters (supplied in the kit) to the DNA ends was achieved by means of the NEBNext Quick Ligation Module (E6056, New England Biolabs, Ipswich, MA, United States).
After each step of library preparation, the sample cleanup was performed using the AMPure XP beads (Agentcourt, Beckman). A flow cell quality control (SpotON Flow Cell Mk I R9 Version, FLO-Min 106D) was run prior to the start of sequencing. The MinKNOWTM software was used to check the number of active pores in the flow cell. Finally, the priming and the loading of the DNA library into the flow cell were performed following protocol recommendations.
The Nanopore device (MinION) was connected to a portable computer, and the software MinKNOW was launched after the appropriate protocol script was selected. The sequencing run was performed for 36 h.
Data Analyses
The quality of the run was monitored by checking the duty time plot showing the distribution of channel states—sequencing, pore, recovering, inactive, and unclassified—over time. The resulting fastq data files were base-called live using the Nanopore GPU with the GUPPY base-calling software 3.0.3 on UBUNTU 16.04 (Nanopore Community Platform). GUPPY is a data processing toolkit that contains the Oxford Nanopore Technologies’ base-calling algorithms and several bioinformatic post-processing features. The fastq files generated in the sequencing run were compared with databases using one of the available pipelines for data analyses of the Nanopore Community Platform, following the steps recommended by manufacturers. The selected workflow chosen was “What’s in my pot” (WIMP), which is an EPI2ME workflow for taxonomic classification of base-called sequences (reads) generated by Nanopore sequencing. WIMP initially filters fastq files with a mean q-score below a minimum threshold (defaults to 7). For reads above the quality threshold, the Centrifuge classification engine is executed to assign each read to a taxon in the NCBI taxonomy. The Centrifuge classification results are then filtered and aggregated to calculate and report counts of reads at the species rank. For reads without a reliable assignment at the species rank, higher ranks of the taxonomy tree are used for the assignment. If no placement is reliable enough (below a scoring threshold), the sequence is labeled as “Unclassified.” The run report, including identification data and associated number of reads presented as NCBI taxonomy trees, is accessible in the EPI2ME section of the Nanopore community website.
Results and Discussion
Identification of Polyester-Degrading Activities
Despite the potential of microbial communities from different environments to contain novel polyester-degrading enzymes (Danso et al., 2019), the plenitude of polyester-degrading enzyme activities present in rumen has not yet been fully exploited. Yet rumen from alpine pasture-fed animals could contain polyester-degrading enzymes needed for the digestion of polyesters such as cutin from berries (Kallio et al., 2006).
The protein concentration in rumen samples was 2.5 mg ml–1 (±0.1 mg ml–1). All short-chain p-NP-esters were hydrolyzed into p-nitrophenol, with slightly higher activity on those esters based on C4, C6, and C8 acids (6.5, 5.8, and 5.5 U ml–1, respectively) (Figure 1). Therefore, rumen content demonstrated considerably higher esterase activity in comparison with polyester-induced fungal supernatants, where esterase activity accounted for approximately 0.4 U ml–1 with p-NPB (C4) as a model substrate (Weinberger et al., 2020).
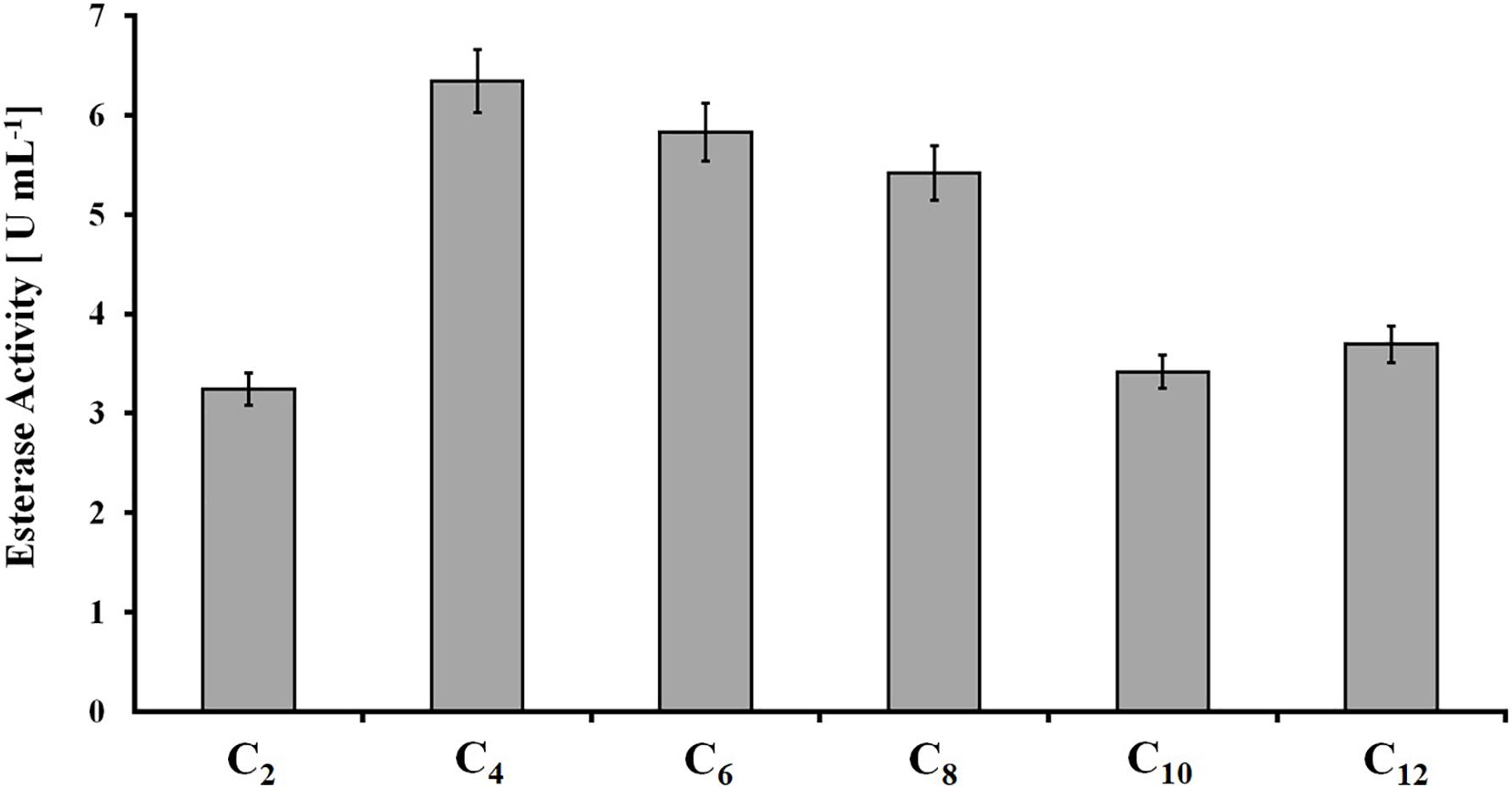
Figure 1. Hydrolysis of para-nitrophenyl-esters with different fatty acid chain lengths (C2 to C12) by rumen samples. Data are mean values of triplicate experiments, and bars represent the standard deviation.
The investigation of hydrolysis efficiencies revealed that rumen samples were able to degrade all three tested polyesters. As expected, the hydrolysis of powders was faster in comparison to films, due to the higher surface-to-volume ratio. After 72 h, 0.6 and 0.75 mM of hydrolysis products were released from the aromatic polyesters PET and PBAT, respectively. Considerably higher amounts (4.8 mM) of hydrolysis products (namely, FDCA) were released from PEF (Figure 2).
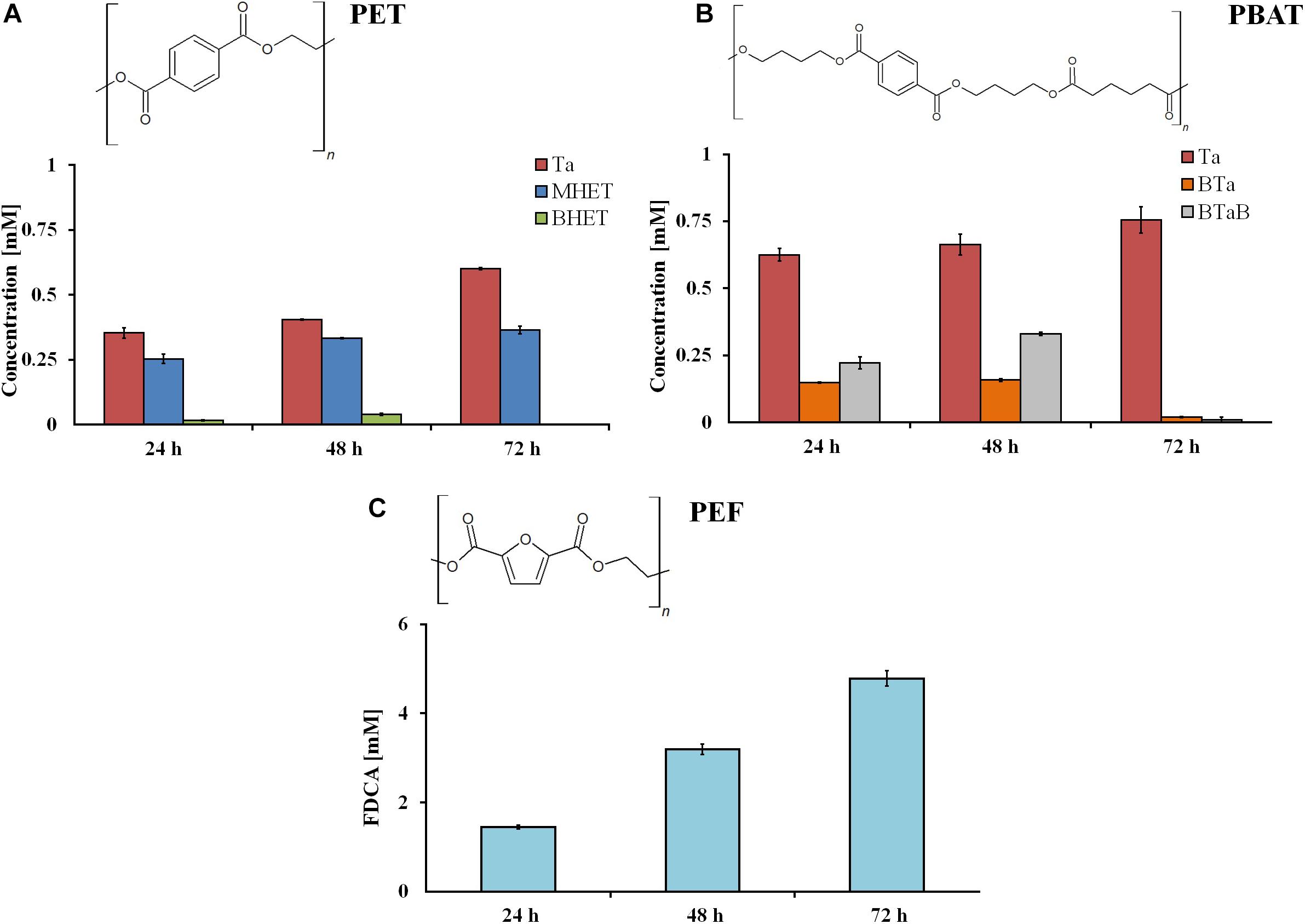
Figure 2. Hydrolysis products after incubation of different synthetic polyester powders (PET (A), PBAT (B), and PEF (C)) with rumen samples. Ta, terephthalic acid; MHET, mono-2-hydroxyethyl terephthalate; BHET, bis(2-hydroxyethyl)terephthalate; BTa, mono(hydroxybutyl)terephthalate; BTaB, bis(4-hydroxybutyl) terephthalate; FDCA, 2.5-furandicarboxylic acid. Data are mean values of three different measurements, and bars represent the standard deviation.
In comparison to polymer powder, much lower amounts of hydrolysis products were released from all polyester films when compared to the powders. After 72 h, 0.15 mM of Ta were measured from PET (Figure 3A), while from PBAT, a higher amount (0.5 mM) of Ta was released most likely due to its aliphatic component. In case of PBAT, it can be clearly seen that oligomers released are subsequently hydrolyzed after prolonged incubation (Figure 3B). Like for the other polyesters, a lower amount of hydrolysis products (0.78 mM) was released from PEF films when compared to powders (Figure 3C). Nevertheless, in case of both films and powders, the rumen enzymes were more active on PEF when compared to PET. Apart from the chemical composition (FDCA versus Ta as acid components), other properties can have a strong influence on enzymatic hydrolysis. The polymer chain must be flexible enough to allow access for and sorption of the enzymes. Furthermore, the degree of crystallinity, chain length, or possible intermolecular bonds can have an impact on hydrolysis (Wei and Zimmermann, 2017).
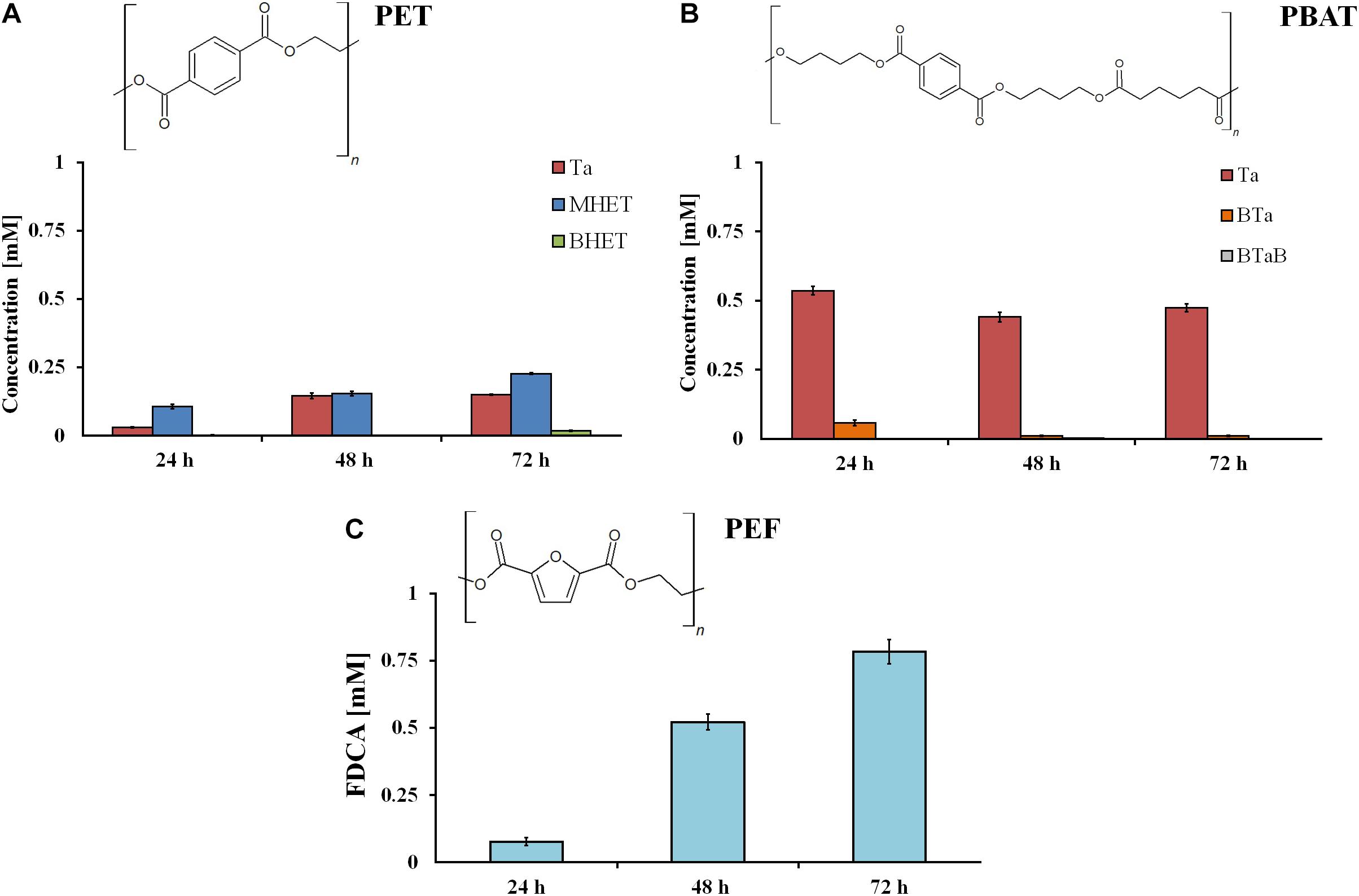
Figure 3. Hydrolysis products after incubation of different synthetic polyester films (PET (A), PBAT (B), and PEF (C)) with rumen samples. Data are mean values of three different measurements, and bars represent the standard deviation.
Since the enzymes are unable to diffuse into the polymer matrix due to their size, hydrolysis starts with surface erosion. While a quite uniform hydrolysis with small cavity formation was observed for PET, both PBAT and PEF formed rougher surfaces already after 1 day. In PBAT films, groove-like structures were seen while for PEF crater-like structures were formed (Figure 4).

Figure 4. Scanning electron microscopy (SEM) of PET, PBAT, and PEF films after 24, 48, and 72 h incubation with rumen samples at 40°C. The left side pictures show washed and untreated films.
Previous studies have investigated the potential of synthetic polyester degradation using metagenomic libraries combined, e.g., with agar plate-based activity assay (Haernvall et al., 2017; Bollinger et al., 2020b; Molitor et al., 2020). This study was designed based on incubation with polymers, in order to assign polyester hydrolysis activity in the rumen microbiome to identified possible candidates within this community. Despite this difference, most of the previous investigations on polyester degradations were conducted using singular microorganisms. For example, Yarrowia lipolytica has been investigated for amorphous PET film degradation, where 0.06 mM Ta was released after 72 h (da Costa et al., 2020). Recently, the black fungus Knufia chersonesos showed the presence of polyesterases, able to cleave PBAT (0.6 mM Ta released), while from enzymatic treatment with rumen, 0.75 mM of the same monomer was released (Tesei et al., 2020). These preliminary results can confirm that different enzymatic activities and/or microorganisms (like rumen microbiome) can break down polymeric structures, as also demonstrated by Barth et al. (2016). On the other hand, PET surface erosion was comparable with previous investigations using different purified variants of the Thermobifida cellulosilytica cutinases 1 and 2 (Thc_Cut1 and Thc_Cut2) (Vecchiato et al., 2017) but was lower when compared with purified engineered PETase from Ideonella sakaiensis, which was able to release 0.5 mM Ta from PET film, whereas 0.15 mM of the monomer was obtained from rumen incubation (Yoshida et al., 2016). Recently, the thermophilic Clostridium thermocellum was engineered for the extracellular secretion of LC cutinase, which resulted in around 1 mM terephthalic acid released (Yan et al., 2021). This is more than that found in this study for the wild-type rumen consortium and indicates the potential of further engineering of promising candidate microbes/enzymes from the rumen consortium.
Microbial Community Analysis
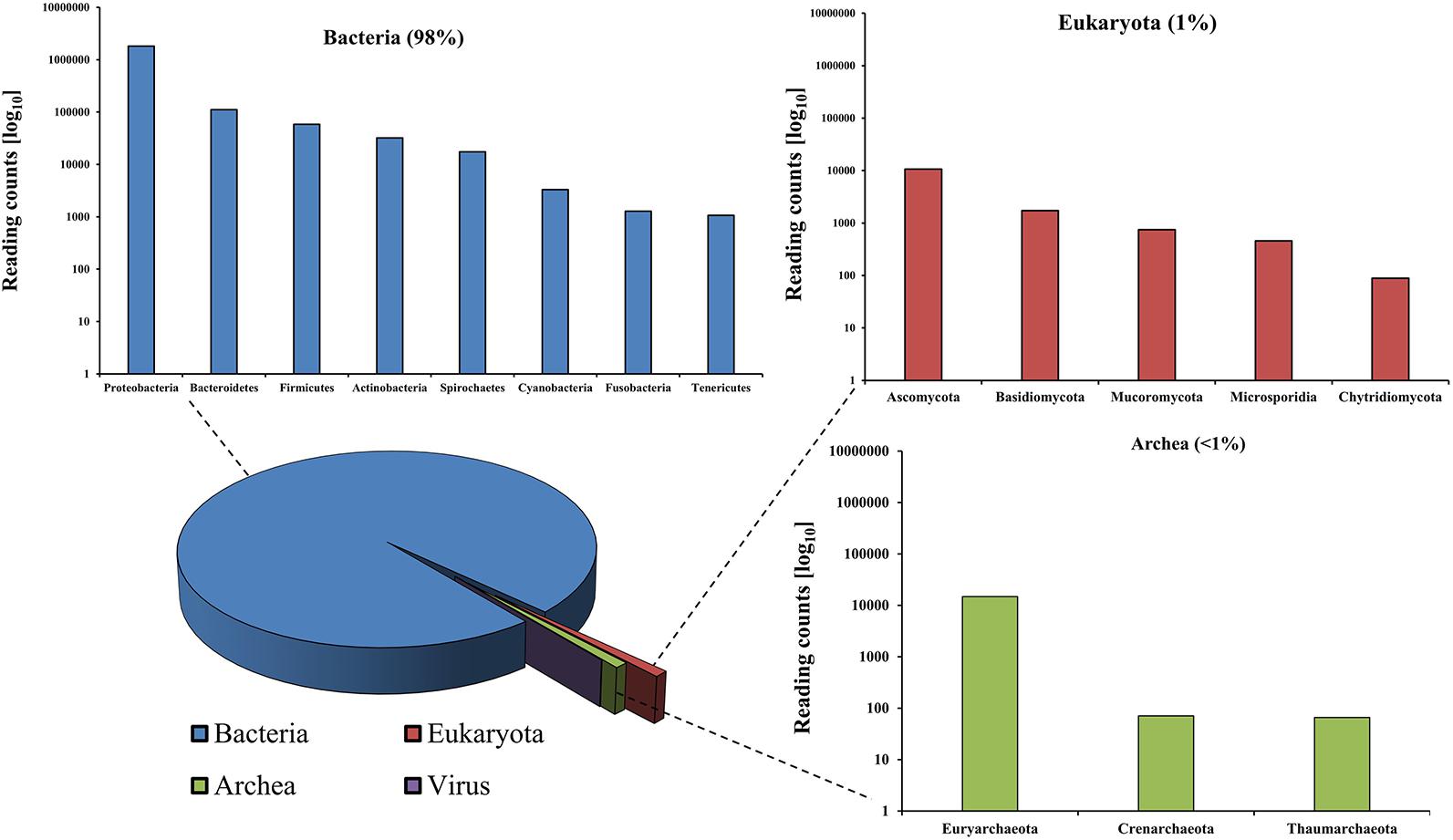
The analysis of extracted DNA from rumen content was performed via shotgun metagenomics using sequencing platform, namely, the MinION Nanopore technology (Brown et al., 2017). Around 3.6 million reads and a total 14.8 Gb of DNA were obtained, with an average length of 4,084 bases (Supplementary Figure 1). More than two million, out of the total number of reads obtained, could be taxonomically classified. The shotgun metagenomic approach applied in this study allowed us to infer the actual proportions of all domains of life and thus an overall identification of the rumen microbial community, which revealed 98% of Bacteria, 1% of Eukaryota, around 0.9% of Archaea, and finally less than 0.1% of viral origin (Figure 5).
Specifically, among the bacterial kingdom, Proteobacteria were predominant, followed by Bacteroidetes, Firmicutes, and Actinobacteria. Species of the Pseudomonas genus accounted for the highest number of reads of all identified genera within the rumen microbiome (Supplementary Figure 2). Pseudomonas spp. are typically present in food and feed and are responsible for enzymatic spoilage (Caldera et al., 2016). Interestingly, Pseudomonas veronii (>106 reads), which is known to be involved in degradation of alkyl methyl ketones (Onaca et al., 2007), was the most dominant species in the rumen microbiome. Acinetobacter was demonstrated to be the second most common species in this microbial community, which is involved in enhancing the intestinal digestive activities and improving feed utilization (Mao et al., 2013).
Rumen Eukaryotes can be mostly considered to belong to the Opisthokonta which comprise metazoans (animals), fungi, and other microbial Eukaryotes, including the Choanoflagellida, Ichthyosporea, Nucleariida, and Capsaspora (Supplementary Figure 3) (Karpov et al., 2014). Fungi comprised Ascomycota and Basidiomycota phyla, which are important for the degradation and the use of fibrous cellulose feed. Another detected eukaryotic phylum was Mucoromycota, which is often associated with plant matter and therefore introduced with the ruminant’s diet.
Expectedly, members of the domain of Archaea, which are responsible for ruminant methanogenesis (Patra et al., 2017), were found, including Euryarchaeota and, within this phylum, methanogenic organisms like members of the genus Methanobrevibacter, which are able to reduce acetic acid into carbon dioxide and methane, as the most abundant genus (Supplementary Figure 4).
Potential Polyester-Degrading Microorganisms
Among hydrolases, esterases, lipases, and cutinases have been described to hydrolyze synthetic polyesters while proteases may hydrolyze polyamides. Indeed, such activities have been reported for abundant species identified in the rumen microbiota (Table 1). Considering the number of generated and taxonomically classified reads in this study, Pseudomonas spp. in rumen samples may play a major role in polyester degradation. These bacteria are well known to be able to produce different hydrolytic activities: esterases, lipases, and cutinases (Bollinger et al., 2020a,b), for all of which hydrolysis of PET has been previously reported. For example, an arylesterase from P. pseudoalcaligenes was recently demonstrated to hydrolyze ionic phthalic polyesters and PBAT (Wallace et al., 2017) while enzymatic polyester-polyurethane hydrolysis was reported for Pseudomonas fluorescens (Jia et al., 2021). On the other hand, P. veronii has been characterized for potential application toward bioremediation of crude oil, hydrocarbons, and aromatic and other hydrophobic compounds (McCarthy et al., 2017). Moreover, different bacterial species (like Pseudomonas) can utilize polyester hydrolysis products such as terephthalic acid as carbon sources (Wilkes and Aristilde, 2017). Acinetobacter spp. are well-known bacteria able to utilize different hydrophobic compounds as nutrients, including triglycerides and waxy esters. Therefore, in different studies, Acinetobacter spp. were successfully used for biodegradation of synthetic polyesters in the environment (Dong et al., 2017).
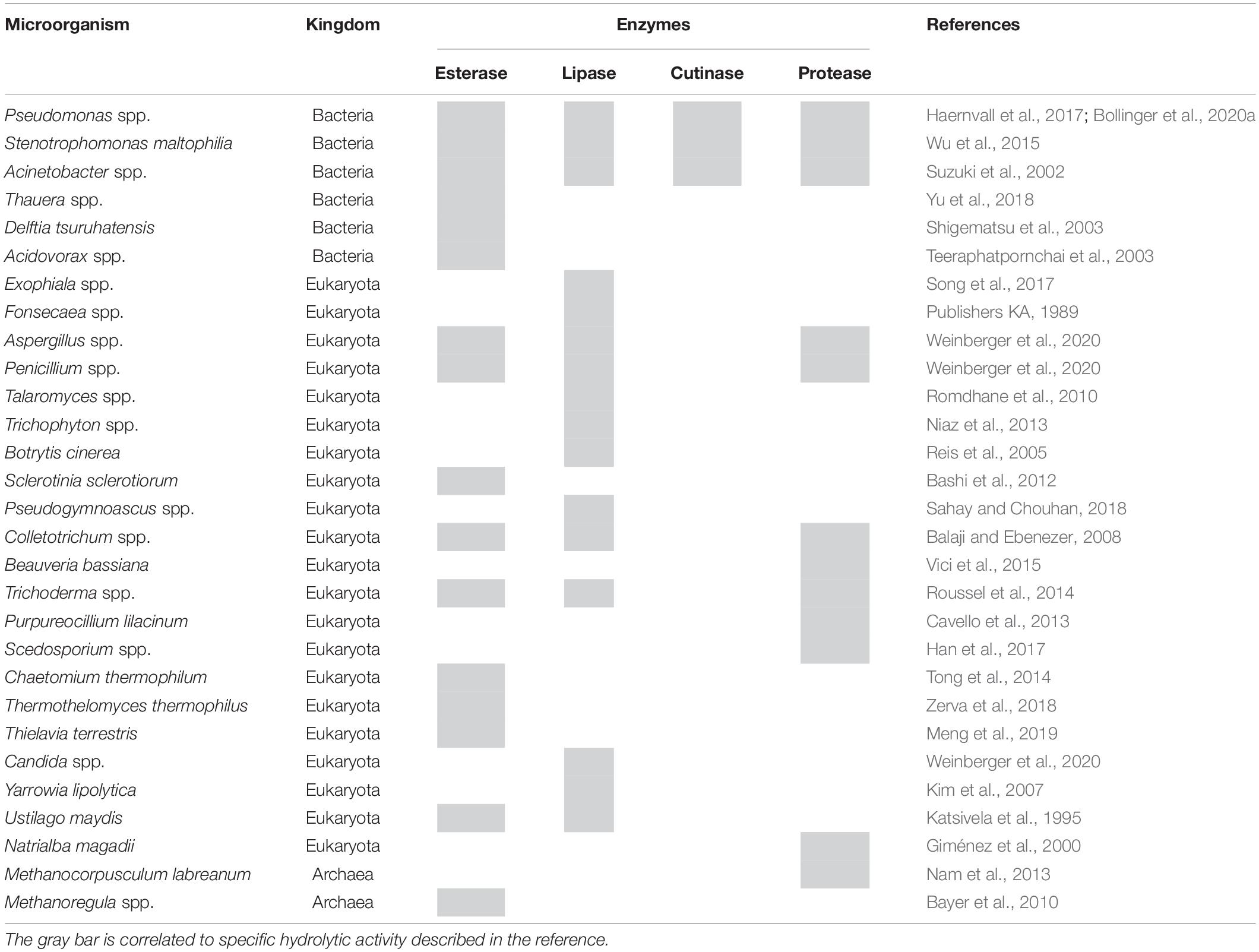
Table 1. Analysis of the rumen microbial community related to the production of enzymes potentially capable of polyester degradation.
Regarding fungi, different hydrolases (lipase and esterase) from Aspergillus, Penicillium, and Candida species are already exploited at industrial level and found applications in different sectors like detergents, textile industry, diary, and food preparation (Weinberger et al., 2020). Hydrolysis of polyesters and other synthetic polymers was previously described for Penicillium and Beauveria species (Heumann et al., 2006; Liebminger et al., 2007; Almansa et al., 2009). Fungi involved in plant cell wall degradation, like Thermothelomyces thermophilus, Talaromyces wortmannii (Zerva et al., 2018), and Thielavia terrestris (Meng et al., 2019), produce feruloyl esterases. This class of hydrolases cleaves the ester bonds between hydroxycinnamic acid esterified with arabinoxylans (Wong, 2006). As shown by Tesei et al. (2020) the black fungus K. chersonesos is able to secrete an esterase which shows high similarity with either a feruloyl esterase (i.e., AoFaeB from Aspergillus oryzae) or MHETase from I. sakaiensis, suggesting therefore that it can be possible that this class of hydrolase is involved in synthetic polyester degradation.
On the other hand, enzymes from Archaea are generally under-explored for biotechnological applications. Since the microorganisms belonging to this domain can live and tolerate extreme conditions (e.g., high temperature, salinity, alkalinity, or acidity), identification of polyester-degrading enzymes could have interesting properties for industrial applications. Among the Archaea identified in the rumen samples, thermophilic esterases have been described for Methanocorpusculum labreanum and Methanoregula sp. (Bayer et al., 2010; Nam et al., 2013).
As a proof of principle, the activity of selected microorganisms (i.e., their enzymes) listed in Table 1 was assessed. Among fungi, indeed P. citrinum (i.e., culture filtrate) was able to hydrolyze PET while no significant activity was found for B. bassiana in terms of release of Ta. Among bacteria, P. pseudoalcaligenes was able to hydrolyze aromatic polyesters.
Conclusion
The rumen microbial community has demonstrated the ability to degrade three different synthetic polyesters (PET, PBAT, and PEF) as demonstrated by quantification of solubilized molecules and SEM analysis of surface erosion. In nature, polyester-hydrolyzing activity of rumen microbes may be involved in the digestion of polyesters, such as cutin present in the diet of ruminants. Among the bacteria, fungi and archaea identified in this study by microbial community analysis as two of the most abundant species have been described to produce enzymes potentially capable of polyester hydrolysis. Compared to published data for pure enzymes and/or supernatants of single microorganisms, the polyester-hydrolyzing activity of rumen fluid was relatively high. Apparently, not only one type of enzymes is present in the rumen mixture, but rather a synergistic action of different esterases, lipases, or cutinases may occur.
This is not surprising since there are many examples in nature of biopolymers (e.g., cellulose) where different enzymes are involved in synergistic degradation. Despite the fact that rumen fluid could be a cheap source for polymer-degrading enzymes, future studies should aim at identification and cultivation of the microbes and enzymes involved in synergistic hydrolysis of polyesters as well as on possible community changes during incubation with polyesters.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
Author Contributions
FQ, KK, and HS performed the experiments. FQ, DR, and GG planned the experiments. LP performed the SEM measurements. DT, GP, and KS performed the DNA extraction and MinION measurement. FQ and DR wrote the manuscript. DR and GG supervised the work. All authors discussed the collected data and corrected the manuscript before submission.
Funding
The COMET center: acib: Next Generation Bioproduction was funded by the BMVIT, BMDW, SFG, Standortagentur Tirol, Government of Lower Austria, and Vienna Business Agency in the framework of COMET—Competence Centers for Excellent Technologies. The COMET-Funding Program is managed by the Austrian Research Promotion Agency FFG. Open access funding provided by BOKU Vienna Open Access Publishing Fund.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fbioe.2021.684459/full#supplementary-material
References
Almansa, E., Heumann, S., Eberl, A., and Kaufmann, F. (2009). Surface hydrolysis of polyamide with a new polyamidase from Beauveria brongniartii. Biocatal. Biotransformation 26, 371–377. doi: 10.1080/10242420802323433
Balaji, V., and Ebenezer, P. (2008). Optimization of extracellular lipase production in Colletotrichum gloeosporioides by solid state fermentation. Indian J. Sci. Technol. 1, 1–8.
Barth, M., Honak, A., Oeser, T., Wei, R., Belisário-Ferrari, M. R., Then, J., et al. (2016). A dual enzyme system composed of a polyester hydrolase and a carboxylesterase enhances the biocatalytic degradation of polyethylene terephthalate films. Biotechnol. J. 11, 1082–1087. doi: 10.1002/biot.201600008
Bashi, Z. D., Roger Rimmer, S., Khachatourians, G. G., and Hegedus, D. D. (2012). Factors governing the regulation of Sclerotinia sclerotiorum cutinase a and polygalacturonase 1 during different stages of infection. Can. J. Microbiol. 58, 605–616. doi: 10.1139/w2012-031
Bayer, S., Kunert, A., Ballschmiter, M., and Greiner-Stoeffele, T. (2010). Indication for a new lipolytic enzyme family: isolation and characterization of two esterases from a metagenomic library. J. Mol. Microbiol. Biotechnol. 18, 181–187. doi: 10.1159/000315459
Biundo, A., Hromic, A., Pavkov-Keller, T., Gruber, K., Quartinello, F., Haernvall, K., et al. (2016). Characterization of a poly(butylene adipate-co-terephthalate)-hydrolyzing lipase from Pelosinus fermentans. Appl. Microbiol. Biotechnol. 100, 1753–1764. doi: 10.1007/s00253-015-7031-1
Biundo, A., Ribitsch, D., and Guebitz, G. M. (2018). Surface engineering of polyester-degrading enzymes to improve efficiency and tune specificity. Appl. Microbiol. Biotechnol. 102, 3551–3559. doi: 10.1007/s00253-018-8850-7
Bollinger, A., Thies, S., Katzke, N., and Jaeger, K. E. (2020a). The biotechnological potential of marine bacteria in the novel lineage of Pseudomonas pertucinogena. Microb. Biotechnol. 13, 19–31. doi: 10.1111/1751-7915.13288
Bollinger, A., Thies, S., Knieps-Grünhagen, E., Gertzen, C., Kobus, S., Höppner, A., et al. (2020b). A novel polyester hydrolase from the marine bacterium Pseudomonas aestusnigri – structural and functional insights. Front. Microbiol. 11:114. doi: 10.3389/fmicb.2020.00114
Brown, B. L., Watson, M., Minot, S. S., Rivera, M. C., and Franklin, R. B. (2017). MinIONTM nanopore sequencing of environmental metagenomes: a synthetic approach. Gigascience 6, 1–10. doi: 10.1002/9783527678679.dg08196
Caldera, L., Franzetti, L., van Coillie, E., De Vos, P., Stragier, P., De Block, J., et al. (2016). Identification, enzymatic spoilage characterization and proteolytic activity quantification of Pseudomonas spp. isolated from different foods. Food Microbiol. 54, 142–153. doi: 10.1016/j.fm.2015.10.004
Cavello, I. A., Hours, R. A., Rojas, N. L., and Cavalitto, S. F. (2013). Purification and characterization of a keratinolytic serine protease from Purpureocillium lilacinum LPS # 876. Process Biochem. 48, 972–978. doi: 10.1016/j.procbio.2013.03.012
da Costa, A. M., de Oliveira Lopes, V. R., Vidal, L., Nicaud, J. M., de Castro, A. M., and Coelho, M. A. Z. (2020). Poly(ethylene terephthalate) (PET) degradation by Yarrowia lipolytica: investigations on cell growth, enzyme production and monomers consumption. Process Biochem. 95, 81–90. doi: 10.1016/j.procbio.2020.04.001
Danso, D., Chow, J., and Wolfgang, R. S. (2019). Plastics?: environmental and biotechnological perspectives on microbial degradation. Appl. Environ. Microbiol. 85:e01095-19. doi: 10.1128/AEM.01095-19
Song, Y., Laureijssen-van de Sande, W. W. J., Moreno, L. F., Gerrits van den Ende, B., Li, R., and de Hoog, S. (2017). Comparative Ecology of Capsular Exophiala Species Causing Disseminated Infection in Humans. Front Microbiol. 8:2514. doi: 10.3389/fmicb.2017.02514
Dong, H., Secundo, F., Xue, C., and Mao, X. (2017). Whole-cell biocatalytic synthesis of cinnamyl acetate with a novel esterase from the DNA library of Acinetobacter hemolyticus. J. Agric. Food Chem. 65, 2120–2128. doi: 10.1021/acs.jafc.6b05799
Gijzen, H. J., Lubberding, H. J., Verhagen, F. J., Zwart, K. B., and Vogels, G. D. (1987). Application of rumen microorganisms for an enhanced anaerobic degradation of solid organic waste materials. Biol. Wastes 22, 81–95. doi: 10.1016/0269-7483(87)90041-3
Giménez, M. I., Studdert, C. A., Sánchez, J. J., and De Castro, R. E. (2000). Extracellular protease of Natrialba magadii: purification and biochemical characterization. Extremophiles 4, 181–188. doi: 10.1007/s007920070033
Haernvall, K., Zitzenbacher, S., Wallig, K., Yamamoto, M., Schick, M. B., Ribitsch, D., et al. (2017). Hydrolysis of ionic phthalic acid based polyesters by wastewater microorganisms and their enzymes. Environ. Sci. Technol. 51, 4596–4605. doi: 10.1021/acs.est.7b00062
Han, Z., Kautto, L., and Nevalainen, H. (2017). Secretion of proteases by an opportunistic fungal pathogen Scedosporium aurantiacum. PLoS One 12:e0169403. doi: 10.1371/journal.pone.0169403
Herrero Acero, E., Ribitsch, D., Steinkellner, G., Gruber, K., Greimel, K., Eiteljoerg, I., et al. (2011). Enzymatic surface hydrolysis of PET: effect of structural diversity on kinetic properties of cutinases from Thermobifida. Macromolecules 44, 4632–4640. doi: 10.1021/ma200949p
Heumann, S., Eberl, A., Pobeheim, H., Liebminger, S., Fischer-colbrie, G., and Almansa, E. (2006). New model substrates for enzymes hydrolysing polyethyleneterephthalate and polyamide fibres. J. Biochem. Biophys. Methods 39, 89–99. doi: 10.1016/j.jbbm.2006.02.005
Jia, H., Zhang, M., Weng, Y., and Li, C. (2021). Degradation of polylactic acid/polybutylene adipate-co-terephthalate by coculture of Pseudomonas mendocina and Actinomucor elegans. J. Hazard. Mater. 403:123679. doi: 10.1016/j.jhazmat.2020.123679
Kallio, H., Nieminen, R., Tuomasjukka, S., and Hakala, M. (2006). Cutin composition of five finnish berries. J. Agric. Food Chem. 54, 457–462. doi: 10.1021/jf0522659
Karpov, S. A., Mamkaeva, M. A., Aleoshin, V. V., Nassonova, E., Lilje, O., and Gleason, F. H. (2014). Morphology, phylogeny, and ecology of the aphelids (Aphelidea, Opisthokonta) and proposal for the new superphylum Opisthosporidia. Front. Microbiol. 5:112. doi: 10.3389/fmicb.2014.00112
Katsivela, E., Kleppe, F., Lang, S., and Wagner, F. (1995). Ustilago maydis lipase I. Hydrolysis and ester-synthesis activities of crude enzyme preparation. Enzyme Microb. Technol. 17, 739–745. doi: 10.1016/0141-0229(94)00127-d
Kim, J. T., Kang, S. G., Woo, J. H., Lee, J. H., Jeong, B. C., and Kim, S. J. (2007). Screening and its potential application of lipolytic activity from a marine environment: characterization of a novel esterase from Yarrowia lipolytica CL180. Appl. Microbiol. Biotechnol. 74, 820–828. doi: 10.1007/s00253-006-0727-5
Liebminger, S., Eberl, A., Sousa, F., Heumann, S., Fischer-Colbrie, G., Cavaco-Paulo, A., et al. (2007). Hydrolysis of PET and bis-(benzoyloxyethyl) terephthalate with a new polyesterase from Penicillium citrinum. Biocatal. Biotransformation 25, 171–177. doi: 10.1080/10242420701379734
Mao, S. Y., Zhang, R. Y., Wang, D. S., and Zhu, W. Y. (2013). Impact of subacute ruminal acidosis (SARA) adaptation on rumen microbiota in dairy cattle using pyrosequencing. Anaerobe 24, 12–19. doi: 10.1016/j.anaerobe.2013.08.003
McCarthy, D., Edwards, G. C., Gustin, M. S., Care, A., Miller, M. B., and Sunna, A. (2017). An innovative approach to bioremediation of mercury contaminated soils from industrial mining operations. Chemosphere 184, 694–699. doi: 10.1016/j.chemosphere.2017.06.051
Meng, Z., Yang, Q. Z., Wang, J. Z., and Hou, Y. H. (2019). Cloning, characterization, and functional expression of a thermostable type B feruloyl esterase from thermophilic Thielavia terrestris. Appl. Biochem. Biotechnol. 189, 1304–1317. doi: 10.1007/s12010-019-03065-3
Molitor, R., Bollinger, A., Kubicki, S., Loeschcke, A., Jaeger, K. E., and Thies, S. (2020). Agar plate-based screening methods for the identification of polyester hydrolysis by Pseudomonas species. Microb. Biotechnol. 13, 274–284. doi: 10.1111/1751-7915.13418
Müller, C. A., Perz, V., Provasnek, C., Quartinello, F., Guebitz, G. M., and Berg, G. (2017). Discovery of polyesterases from moss-associated microorganisms. Appl. Environ. Microbiol. 83:e02641-16.
Nam, J. K., Park, Y. J., and Lee, H. B. (2013). Cloning, expression, purification, and characterization of a thermostable esterase from the archaeon Sulfolobus solfataricus P1. J. Mol. Catal. B Enzym. 94, 95–103. doi: 10.1016/j.molcatb.2013.05.019
Niaz, M., Iftikhar, T., Akram, F., and Niaz, M. (2013). Protocol optimization for extracellular lipase production by trichophyton spp. (MBL 23) under solid state fermentation. Pakistan J. Bot. 45, 1417–1421.
Onaca, C., Kieninger, M., Engesser, K. H., and Altenbuchner, J. (2007). Degradation of alkyl methyl ketones by Pseudomonas veronii MEK700. J. Bacteriol. 189, 3759–3767. doi: 10.1128/jb.01279-06
Patra, A., Park, T., Kim, M., and Yu, Z. (2017). Rumen methanogens and mitigation of methane emission by anti-methanogenic compounds and substances. J. Anim. Sci. Biotechnol. 8:13.
Pellis, A., Gamerith, C., Ghazaryan, G., Ortner, A., Herrero Acero, E., and Guebitz, G. M. (2016a). Ultrasound-enhanced enzymatic hydrolysis of poly(ethylene terephthalate). Bioresour. Technol. 218, 1298–1302. doi: 10.1016/j.biortech.2016.07.106
Pellis, A., Herrero Acero, E., Ferrario, V., Ribitsch, D., Guebitz, G. M., and Gardossi, L. (2016b). The closure of the cycle: enzymatic synthesis and functionalization of bio-based polyesters. Trends Biotechnol. 34, 316–328. doi: 10.1016/j.tibtech.2015.12.009
Perz, V., Baumschlager, A., Bleymaier, K., Zitzenbacher, S., Hromic, A., Steinkellner, G., et al. (2016). Hydrolysis of synthetic polyesters by Clostridium botulinum esterases. Biotechnol. Bioeng. 113, 1024–1034.
Pinnell, L. J., and Turner, J. W. (2019). Shotgun metagenomics reveals the benthic microbial community response to plastic and bioplastic in a coastal marine environment. Front. Microbiol. 10:1252.
Plastics Europe, and EPRO (2019). Plastics - the Facts 2019. Available online at: https://www.plasticseurope.org/en/resources/market-data (accessed March, 2021).
Publishers KA (1989). Lipases of Fonsecaea pedrosoi and Phialophora verrucosa lipase production lipase assay the lipase activity was estimated titrimetrically (Somkuti & Babel 1968). One 324, 313–324. doi: 10.1007/bf00398510
Quartinello, F., Kremser, K., Vecchiato, S., Schoen, H., Vielnascher, R., Ploszczanski, L., et al. (2019). Increased flame retardancy of enzymatic functionalized PET and nylon fabrics via DNA immobilization. Front. Chem. 7:685. doi: 10.3389/fchem.2019.00685
Quartinello, F., Vajnhandl, S., Volmajer Valh, J., Farmer, T. J., Vončina, B., Lobnik, A., et al. (2017). Synergistic chemo-enzymatic hydrolysis of poly(ethylene terephthalate) from textile waste. Microb. Biotechnol. 10, 1376–1383. doi: 10.1111/1751-7915.12734
Reis, H., Pfiff, S., and Hahn, M. (2005). Molecular and functional characterization of a secreted lipase from Botrytis cinerea. Mol. Plant Pathol. 6, 257–267. doi: 10.1111/j.1364-3703.2005.00280.x
Ribitsch, D., Yebra, A. O., Zitzenbacher, S., Wu, J., Nowitsch, S., Steinkellner, G., et al. (2013). Fusion of binding domains to Thermobifida cellulosilytica cutinase to tune sorption characteristics and enhancing PET hydrolysis. Biomacromolecules 14, 1769–1776. doi: 10.1021/bm400140u
Romdhane, I. B. B., Fendri, A., Gargouri, Y., Gargouri, A., and Belghith, H. (2010). A novel thermoactive and alkaline lipase from Talaromyces thermophilus fungus for use in laundry detergents. Biochem. Eng. J. 53, 112–120. doi: 10.1016/j.bej.2010.10.002
Roussel, A., Amara, S., Nyyssölä, A., Mateos-Diaz, E., Blangy, S., Kontkanen, H., et al. (2014). A cutinase from Trichoderma reesei with a lid-covered active site and kinetic properties of true lipases. J. Mol. Biol. 426, 3757–3772. doi: 10.1016/j.jmb.2014.09.003
Sahay, S., and Chouhan, D. (2018). Study on the potential of cold-active lipases from psychrotrophic fungi for detergent formulation. J. Genet. Eng. Biotechnol. 16, 319–325. doi: 10.1016/j.jgeb.2018.04.006
Shigematsu, T., Yumihara, K., Ueda, Y., Numaguchi, M., Morimura, S., and Kida, K. (2003). Delftia tsuruhatensis sp. nov., a terephthalate-assimilating bacterium isolated from activated sludge. Int. J. Syst. Evol. Microbiol. 53, 1479–1483. doi: 10.1099/ijs.0.02285-0
Sulaiman, S., Yamato, S., Kanaya, E., Kim, J. J., Koga, Y., Takano, K., et al. (2012). Isolation of a novel cutinase homolog with polyethylene terephthalate-degrading activity from leaf-branch compost by using a metagenomic approach. Appl. Environ. Microbiol. 78, 1556–1562. doi: 10.1128/aem.06725-11
Suzuki, T., Nakayama, T., Kurihara, T., Nishino, T., and Esaki, N. (2002). A cold-active esterase with a substrate preference for vinyl esters from a psychrotroph, Acinetobacter sp. strain no. 6: gene cloning, purification, and characterization. J. Mol. Catal. B Enzym. 16, 255–263. doi: 10.1016/s1381-1177(01)00070-4
Teeraphatpornchai, T., Nakajima-Kambe, T., Shigeno-Akutsu, Y., Nakayama, M., Nomura, N., Nakahara, T., et al. (2003). Isolation and characterization of a bacterium that degrades various polyester-based biodegradable plastics. Biotechnol. Lett. 25, 23–28.
Tesei, D., Quartinello, F., Guebitz, G. M., Ribitsch, D., Nöbauer, K., Razzazi-Fazeli, E., et al. (2020). Shotgun proteomics reveals putative polyesterases in the secretome of the rock-inhabiting fungus Knufia chersonesos. Sci. Rep. 10:9770.
Tong, X., Lange, L., Grell, M. N., and Busk, P. K. (2014). Hydrolysis of wheat arabinoxylan by two acetyl xylan esterases from Chaetomium thermophilum. Appl. Biochem. Biotechnol. 175, 1139–1152. doi: 10.1007/s12010-014-1348-6
Vecchiato, S., Ahrens, J., Pellis, A., Scaini, D., Mueller, B., Herrero Acero, E., et al. (2017). Enzymatic functionalization of HMLS-polyethylene terephthalate fabrics improves the adhesion to rubber. ACS Sustain. Chem. Eng. 5, 6456–6465. doi: 10.1021/acssuschemeng.7b00475
Vici, A. C., da Cruz, A. F., Facchini, F. D. A., de Carvalho, C. C., Pereira, M. G., Fonseca-Maldonado, R., et al. (2015). Beauveria bassiana lipase A expressed in Komagataella (Pichia) pastoris with potential for biodiesel catalysis. Front. Microbiol. 6:1083. doi: 10.3389/fmicb.2015.01083
Wallace, P. W., Haernvall, K., Ribitsch, D., Zitzenbacher, S., Schittmayer, M., Steinkellner, G., et al. (2017). PpEst is a novel PBAT degrading polyesterase identified by proteomic screening of Pseudomonas pseudoalcaligenes. Appl. Microbiol. Biotechnol. 101, 2291–2303. doi: 10.1007/s00253-016-7992-8
Wei, R., and Zimmermann, W. (2017). Microbial enzymes for the recycling of recalcitrant petroleum-based plastics: how far are we? Microb. Biotechnol. 10, 1308–1322. doi: 10.1111/1751-7915.12710
Weinberger, S., Beyer, R., Schüller, C., Strauss, J., Pellis, A., Ribitsch, D., et al. (2020). High throughput screening for new fungal polyester hydrolyzing enzymes. Front. Microbiol. 11:554. doi: 10.3389/fmicb.2020.00554
Weinberger, S., Canadell, J., Quartinello, F., Yeniad, B., Arias, A., Pellis, A., et al. (2017a). Enzymatic degradation of poly(ethylene 2,5-furanoate) powders and amorphous films. Catalysts 7:318. doi: 10.3390/catal7110318
Weinberger, S., Haernvall, K., Scaini, D., Ghazaryan, G., Zumstein, M. T., Sander, M., et al. (2017b). Enzymatic surface hydrolysis of poly(ethylene furanoate) thin films of various crystallinities. Green Chem. 19, 5381–5384. doi: 10.1039/c7gc02905e
Wilkes, R. A., and Aristilde, L. (2017). Degradation and metabolism of synthetic plastics and associated products by Pseudomonas sp.: capabilities and challenges. J. Appl. Microbiol. 123, 582–593. doi: 10.1111/jam.13472
Wong, D. W. S. (2006). Feruloyl esterase: a key enzyme in biomass degradation. Appl. Biochem. Biotechnol. 133, 87–112. doi: 10.1385/abab:133:2:87
Wu, J. P., Li, M., Zhou, Y., Yang, L. R., and Xu, G. (2015). Introducing a salt bridge into the lipase of Stenotrophomonas maltophilia results in a very large increase in thermal stability. Biotechnol. Lett. 37, 403–407. doi: 10.1007/s10529-014-1683-2
Yan, F., Wei, R., Cui, Q., Bornscheuer, U. T., and Liu, Y. J. (2021). Thermophilic whole-cell degradation of polyethylene terephthalate using engineered Clostridium thermocellum. Microb. Biotechnol. 14, 374–385. doi: 10.1111/1751-7915.13580
Yoshida, S., Hiraga, K., Takanaha, T., Taniguchi, I., Yamaji, H., Maeda, Y., et al. (2016). A bacterium that degrades and assimilates poly(ethyleneterephthalate). Science 351, 1196–1199. doi: 10.1126/science.aad6359
Yu, N., Yang, J. C., Yin, G. T., Li, R. S., Zou, W. T., and He, C. (2018). Identification and characterization of a novel esterase from Thauera sp. Biotechnol. Appl. Biochem. 65, 748–755.
Keywords: synthetic polyesters hydrolysis, recycling, microbial communities, rumen, polyester degrading enzymes
Citation: Quartinello F, Kremser K, Schoen H, Tesei D, Ploszczanski L, Nagler M, Podmirseg SM, Insam H, Piñar G, Sterflingler K, Ribitsch D and Guebitz GM (2021) Together Is Better: The Rumen Microbial Community as Biological Toolbox for Degradation of Synthetic Polyesters. Front. Bioeng. Biotechnol. 9:684459. doi: 10.3389/fbioe.2021.684459
Received: 23 March 2021; Accepted: 20 May 2021;
Published: 02 July 2021.
Edited by:
Evangelos Topakas, National Technical University of Athens, GreeceReviewed by:
Karl-Erich Jaeger, Heinrich Heine University Düsseldorf, GermanyTjalf De Boer, MicroLife Solutions, Netherlands
Ya-Jun Liu, Qingdao Institute of Bioenergy and Bioprocess Technology (CAS), China
Copyright © 2021 Quartinello, Kremser, Schoen, Tesei, Ploszczanski, Nagler, Podmirseg, Insam, Piñar, Sterflingler, Ribitsch and Guebitz. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Doris Ribitsch, doris.ribitsch@boku.ac.at
†ORCID: Felice Quartinello, orcid.org/0000-0001-9014-1621; Klemens Kremser, orcid.org/0000-0001-7087-094X; Herta Schoen, orcid.org/0000-0001-8520-5897; Donatella Tesei, orcid.org/000-0002-3247-066X; Leon Ploszczanski, orcid.org/0000-0002-6052-4610; Magdalena Nagler, orcid.org/0000-0002-4165-7290; Sabine M. Podmirseg, orcid.org/0000-0003-1775-2412; Heribert Insam, orcid.org/0000-0002-5136-2752; Guadalupe Piñar, orcid.org/0000-0001-7881-8572; Katja Sterflingler, orcid.org/0000-0002-0296-6728; Doris Ribitsch, orcid.org/0000-0002-5822-0204; Georg M. Guebitz, orcid.org/0000-0003-2262-487X
‡Present address: Guadalupe Piñar and Katja Sterflingler, Academy of Fine Arts Vienna, Institute for Natural Sciences and Technology in the Arts, Vienna, Austria
 Felice Quartinello1,2†
Felice Quartinello1,2†  Donatella Tesei
Donatella Tesei Magdalena Nagler
Magdalena Nagler Sabine M. Podmirseg
Sabine M. Podmirseg Guadalupe Piñar
Guadalupe Piñar Katja Sterflingler
Katja Sterflingler Doris Ribitsch
Doris Ribitsch Georg M. Guebitz
Georg M. Guebitz